The Otto Cycle is the ideal thermodynamic cycle used to describe how spark ignition (SI) engines, commonly known as petrol engines, operate. It was developed by Dr. Nikolaus August Otto, whose work led to the foundation of modern gasoline engines used in motorcycles, cars, and light vehicles.
In simple terms, the Otto cycle explains how fuel energy is converted into mechanical work inside an engine cylinder. It consists of four thermodynamic processes that involve compression, heat addition, expansion, and heat rejection. These processes repeat continuously to produce power.
Understanding the Otto cycle is important because it helps engineers analyze engine efficiency, fuel performance, and power output. It also forms the base for studying other engine cycles like the Diesel cycle and Dual cycle used in internal combustion engines.
Table of Contents
What is the Otto Cycle?
The Otto cycle is an ideal gas power cycle that represents the working principle of spark ignition engines.
It consists of four thermodynamic processes:
- Isentropic Compression
- Constant Volume Heat Addition
- Isentropic Expansion
- Constant Volume Heat Rejection
Among these processes:
- Two are isentropic (reversible adiabatic)
- Two are isochoric (constant volume)
Petrol engines operating on the Otto cycle typically have a compression ratio between 7:1 and 10:1.
Important Thermodynamic Processes in Otto Cycle
Before understanding the cycle, it is important to know two basic thermodynamic processes used in it.
Isentropic Process
An isentropic process is a thermodynamic process in which entropy remains constant.
Characteristics:
- No heat transfer occurs
- The process is reversible
- Also called reversible adiabatic process
In the Otto cycle, both compression and expansion are assumed to be isentropic.
Isochoric Process
An isochoric process occurs when the volume remains constant during the process.
Characteristics:
- Volume does not change
- Pressure and temperature may change
- Heat transfer occurs
In the Otto cycle, heat addition and heat rejection take place at constant volume.
How does Otto Cycle Work?
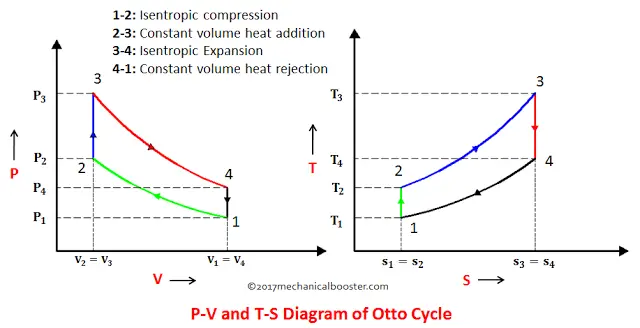
Now, with the help of the P-V and T-S diagram, we can easily understand all the processes of the Otto cycle.

Also Read:
Note: For a better explanation, while reading watch the P-V and T-S diagrams every time.
1. Process 1-2: Isentropic Compression
This process involves the motion of the piston from TDC to BDC. The air that is sucked into the cylinder during suction stroke undergoes reversible adiabatic (isentropic) compression. Since the air is compressed, the pressure increases from P1 to P2, the volume decreases from V1 to V2, the temperature rises from T1 to T2, and entropy remains constant.
2. Process 2-3: Constant Volume Heat Addition
This process is an isochoric process i.e. the heat is added to the air at constant volume. The piston in this process rest for a moment at TDC and during this time heat is added to the air through an external source. Due to the heat addition, the pressure increases from P2 to P3, volume remains constant(i.e. V2=V3), temperature increases from T2 to T3 and entropy increases from S2 to S3.
The amount of heat added is given by

3. Process 3-4: Isentropic Expansion
In this process, the isentropic (reversible adiabatic) expansion of air takes place. The piston moves from TDC to BDC. Power is obtained in this process which is used to do some work. Since this process involves expansion of air, so the pressure decreases from P3 to P4, volume increases from V3 to V4, temperature falls from T3 to T4 and entropy remains unchanged (i.e. S3=S4).
4. Constant Volume Heat Rejection
In this process, the piston rests for a moment at BDC and rejection of heat takes place at constant volume. The pressure decreases from P4 to P1, Volume remains constant (i.e. V4=V1), temperature falls from T4 to T1.
The amount of heat rejected in this process is given by

When this cycle is used in four four-stroke petrol engine than the two process increases. one is the suction of air-fuel mixture inside the cylinder which takes place at constant atmospheric pressure and the other one is the exhaust of gases out of the engine cylinder at constant atmospheric pressure. These two processes are not shown in the ideal Otto cycle that we have discussed above.
Also Read:
Summary in Tabular Form
| S.no | Process | Operation | Position of piston | Change in parameter |
| 1. | 1-2: Isentropic Compression | Compression of air. | BDC to TDC | V: Decreases from V1 To V2 T: Increases from T1 to T2 P: Increases from P1 to P2 S: Entropy remains constant (S1=S2) |
| 2. | 2-3: Constant Volume Heat Addition | Heat is added in the form of spark and combustion occurs. | At TDC for a moment | V: Remains constant (V2 = V3 ) T: Increases from T2 to T3 P: Increases from P2 to P3 S: Increases from S2 to S3 |
| 3. | 3-4: Isentropic Expansion | Expansion of air takes place due to heat addition. | TDC to BDC | V: Increases from V3 to V4 T: Decreases from T3 to T4 P: Decreases from P3 to P4 S: Entropy remains constant (S3=S4) |
| 4. | 4-1: Constant Volume Heat Rejection | Heat is rejected to a sink. | At BDC for a moment | V: Remains constant(V4 = V1 ) T: Decreases from T4 to T1 P: Decreases from P4 to P1 S: Decreases from S4 to S1 |
Thermal Efficiency
The efficiency of Otto cycle is given by

Otto Cycle in a Real Four-Stroke Engine
The ideal Otto cycle describes only four thermodynamic processes. However, in a real four-stroke petrol engine, two additional processes occur:
- Suction stroke – Air-fuel mixture enters the cylinder at atmospheric pressure.
- Exhaust stroke – Burnt gases leave the cylinder.
These processes occur at approximately constant atmospheric pressure and are not included in the ideal Otto cycle.
Application
The Otto cycle is used in spark ignition engines, including:
- Motorcycles
- Cars
- Small petrol generators
- Lawn mowers
- Small aircraft piston engines
- Portable power equipment
These engines are widely used due to their smooth operation, lighter weight, and high speed capability.
Conclusion
The Otto cycle forms the theoretical foundation of modern spark ignition petrol engines. It explains how energy from fuel combustion is converted into mechanical work through four thermodynamic processes: compression, heat addition, expansion, and heat rejection. Understanding the Otto cycle helps engineers evaluate engine performance, improve efficiency, and design better internal combustion engines. Although real engines include additional processes such as suction and exhaust strokes, the Otto cycle remains an essential model for studying petrol engine operation and thermodynamic behavior. Exploring related cycles like the Diesel cycle and Dual cycle can further deepen understanding of engine technology.
The Otto cycle is an ideal thermodynamic cycle used to describe the working of spark ignition petrol engines.
The cycle was developed by Nikolaus August Otto, a German engineer who built the first successful gasoline engine.
The Otto cycle consists of four processes: two isentropic processes and two constant-volume processes.
All petrol engines and spark ignition engines operate on the Otto cycle.
Typical compression ratios range between 7:1 and 10:1 in petrol engines.
It helps engineers understand engine efficiency, power generation, and fuel combustion in petrol engines.
The isentropic expansion (3-4 process) is the power-producing stage.
Heat addition occurs at constant volume during combustion.
The cycle is commonly represented using P-V (Pressure-Volume) and T-S (Temperature-Entropy) diagrams.
Efficiency can be improved by increasing the compression ratio.
Otto Cycle – Knowledge Check
⚠️ Please select an option before continuing.
